CRITICAL CARE TRIALS
Stay ahead of the curve with exciting new clinical trials from the critical care field presented in vivid, visual abstract format. Gain comprehensive and insightful perspectives as each critical development is delivered to you.
PATCH Trauma Trial
Jul 13, 2023
Prehospital Tranexamic Acid for Severe Trauma
This multicenter RCT evaluated prehospital tranexamic acid (TXA) versus placebo in 1310 adults with major trauma at risk for coagulopathy. Survival with favorable functional outcome at 6 months (primary outcome) was similar between groups (53.7% vs. 53.5%; RR 1.00, 95% CI 0.90–1.12, p=0.95). Fewer patients in the TXA group died within 28 days (statistically significant) but not at 6 months. Serious adverse events were comparable. While TXA did not improve long-term functional outcomes, its early mortality benefit supports further exploration in trauma care
TAME Trial
Jul 6, 2023
Mild Hypercapnia or Normocapnia after Out-of-Hospital Cardiac Arrest
This multicenter RCT evaluated targeted mild hypercapnia (PaCO₂ 50–55 mm Hg) versus normocapnia (PaCO₂ 35–45 mm Hg) for 24 hours in 1700 comatose adults resuscitated after out-of-hospital cardiac arrest. At 6 months, favorable neurologic outcomes were similar (43.5% vs. 44.6%; RR 0.98, 95% CI 0.87–1.11, p=0.76), as were mortality rates (48.2% vs. 45.9%; RR 1.05, 95% CI 0.94–1.16). No significant difference in adverse events was observed. These findings support continuing normocapnia as standard care, as mild hypercapnia does not improve neurologic outcomes or
DEVICE Trial
Jun 16, 2023
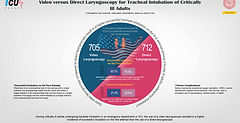
Video versus Direct Laryngoscopy for Tracheal Intubation of Critically Ill Adults
This multicenter RCT compared video laryngoscopy (VL) to direct laryngoscopy (DL) in 1417 critically ill adults undergoing tracheal intubation in emergency departments and ICUs. First-attempt intubation success was higher with VL (85.1% vs. 70.8%; absolute risk difference 14.3%, 95% CI 9.9–18.7, p<0.001). Severe complications were similar between groups. Findings suggest VL improves first-attempt intubation success, particularly for less experienced operators, and supports its routine use in critical care settings.
CAPE-COD
Mar 21, 2023
Hydrocortisone in Severe Community-Acquired Pneumonia
In a phase 3 multicenter RCT of ICU patients with severe community-acquired pneumonia, hydrocortisone significantly reduced 28-day mortality (6.2% vs. 11.9%; absolute difference -5.6%, 95% CI -9.6 to -1.7, P = 0.006). It also lowered rates of intubation (HR 0.59, 95% CI 0.40–0.86) and vasopressor use (HR 0.59, 95% CI 0.43–0.82). Rates of hospital-acquired infections and GI bleeding were similar. Hydrocortisone shows promise as an adjunctive therapy in this setting.
NUTRIREA-3
Mar 20, 2023
Low versus standard calorie and protein feeding in ventilated adults with shock
This multicenter RCT evaluated early low-calorie, low-protein nutrition (6 kcal/kg/day, 0.2–0.4 g/kg/day protein) versus standard targets (25 kcal/kg/day, 1.0–1.3 g/kg/day) in 3036 ICU patients receiving mechanical ventilation and vasopressors for shock. Mortality at 90 days was similar (41.3% vs. 42.8%; p=0.41), but time to ICU discharge readiness was faster in the low group (median 8.0 vs. 9.0 days; HR 1.12, p=0.015). The low group had fewer gastrointestinal complications. Findings suggest a restrictive nutrition strategy may expedite recovery without affecting survival.
BOX Trial (Duration)
Mar 9, 2023
Duration of Device-Based Fever Prevention after Cardiac Arrest
In a randomized trial of comatose patients resuscitated after out-of-hospital cardiac arrest, 36-hour vs. 72-hour device-based fever prevention showed no significant differences in mortality or severe disability/coma. Limitations included unmasked interventions and modest fever prevention efficacy. The results suggest no added benefit to extending device-based fever prevention beyond 36 hours
The CLOVERS Trial
Jan 21, 2023
Early Restrictive or Liberal Fluid Management for Sepsis-Induced Hypotension
In a multicenter trial of 1563 patients with sepsis-induced hypotension, restrictive fluid strategy (early vasopressor use, minimal fluids) showed no significant difference in 90-day mortality compared to liberal fluid strategy (fluid-predominant approach) (14.0% vs. 14.9%; P = 0.61). Secondary outcomes were also similar. Restrictive fluid strategy was safe, with few complications. Results suggest either approach is viable, but findings are limited to early sepsis management.
Spontaneous Breathing Trials
Nov 17, 2022
Spontaneous-Breathing Trials with Pressure-Support Ventilation or a T-Piece
In a multicenter trial of 969 high-risk extubation failure patients, spontaneous-breathing trials with PSV did not significantly increase ventilator-free days at day 28 compared to T-piece trials (median 27 days for both; P = 0.31). Extubation and reintubation rates were similar between groups. Findings suggest that either PSV or T-piece can be used for spontaneous-breathing trials in high-risk patients, with outcomes influenced by prophylactic noninvasive ventilation practices.
AID-ICU
Oct 26, 2022
Haloperidol for the Treatment of Delirium in ICU Patients
In a multicenter RCT of 1000 ICU patients with delirium, haloperidol did not significantly improve days alive and out of the hospital at 90 days compared to placebo (mean 35.8 vs. 32.9 days; adjusted difference 2.9 days; 95% CI -1.2 to 7.0; P = 0.22). Mortality was lower but not statistically significant (36.3% vs. 43.3%). Serious adverse reactions were similar. Haloperidol may aid in managing hyperactive delirium but does not improve long-term outcomes.
The PILOT Trial
Oct 24, 2022
Oxygen-Saturation Targets for Critically Ill Adults Receiving Mechanical Ventilation
In a single-center, cluster-randomized trial of 2541 mechanically ventilated adults, there was no significant difference in ventilator-free days at day 28 between lower (88–92%), intermediate (92–96%), or higher (96–100%) oxygen-saturation targets (median 20 vs. 21 vs. 21 days; P = 0.81). Mortality and adverse events were similar across groups. The findings suggest no clear benefit or harm from varying oxygen saturation targets, supporting flexibility in clinical practice.
HACA-IHCA
Sep 28, 2022
Temperature Control After In-Hospital Cardiac Arrest: A Randomized Clinical Trial
In a multicenter trial of 249 comatose patients after in-hospital cardiac arrest (IHCA), hypothermic temperature control (32–34°C) did not significantly improve 180-day mortality (72.5% vs. 71.2%; RR 1.03, 95% CI 0.79–1.40, P = 0.822) or functional outcomes (22.5% vs. 23.7%; RR 1.04, 95% CI 0.78–1.44, P = 0.822) compared to normothermia (36–37.5°C). While the study was underpowered, it supports maintaining normothermia and avoiding fever in post-IHCA care.
WATERFALL Trial
Sep 15, 2022
Aggressive or Moderate Fluid Resuscitation in Acute Pancreatitis
In a multicenter RCT of 249 patients with acute pancreatitis, aggressive fluid resuscitation did not significantly reduce rates of moderately severe or severe pancreatitis (22.1% vs. 17.3%; RR 1.30, 95% CI 0.78–2.18, P = 0.32) but was associated with higher fluid overload risk (20.5% vs. 6.3%; RR 2.85, 95% CI 1.36–5.94, P = 0.004). The trial was terminated early for safety concerns. Moderate fluid resuscitation is safer and recommended in this population.